Working together
to find a life
without CML

Registration for CML Horizons 2024 Hybrid Conference is NOW OPEN!
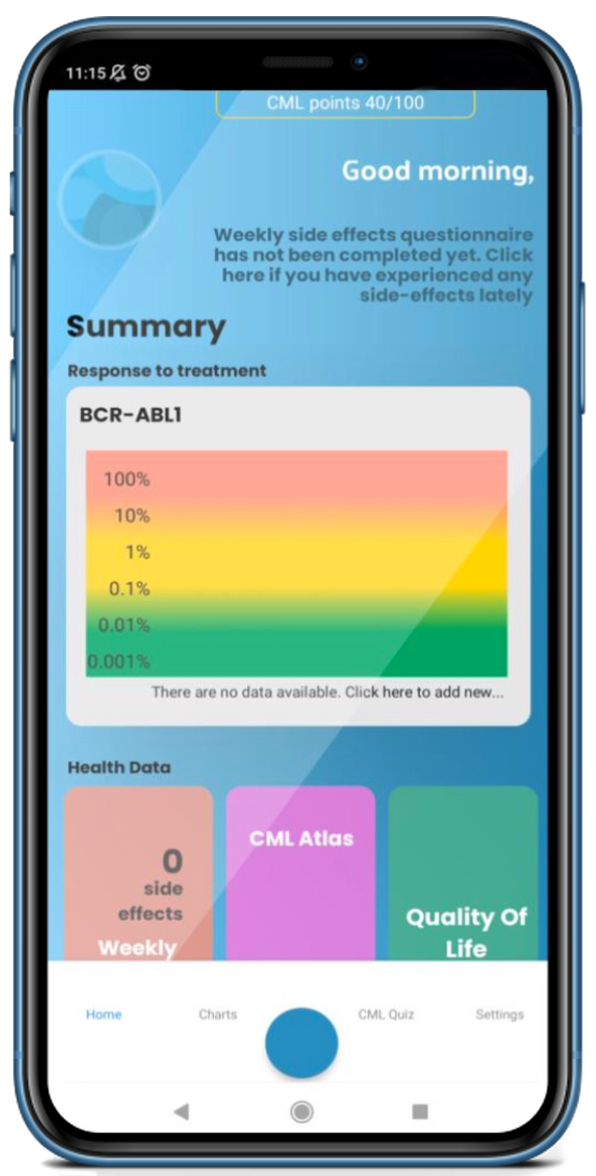
Brighten the way to understanding CML with the Know your CML App
Download today & discover how United we can work to leave no one behind, this World CML Day.
Welcome to the
CML Advocates Network
A network of 131 chronic myeloid leukemia (CML) patient organizations from 96 countries on all continents, the CML Advocates Network serves as a resource for leaders of CML patient groups. As a patient and caregiver-led organization, it was established and is run by patients and caregivers. The organization aims to educate members about advocacy and health equity, empower patient groups to advocate, and implement advocacy initiatives in both low-middle-income (LMI) and western countries. It also aims to facilitate best practices sharing among patient advocates globally.
During the 4th International “New Horizons in Cancer” conference in Dublin in 2005 for patient advocacy groups fighting CML and GIST, the idea for the CML Advocates Network was initially discussed. “New Horizons” conferences originated in 2002 and have become one of the world’s most influential networking and knowledge-sharing events for patient groups dealing with CML and GIST.

Keep yourself informed!
The CML Advocates Network newsletter provides you with information about upcoming CML advocacy events and projects as well as CML-specific news & stories.
The latest news from the network
APPIS is back! Register now for APPIS 2024!
The Alliance and Partnerships for Patient Innovation and Solutions (APPIS) is an organization that aims to bring together individuals and groups to create innovative solutions for patients. With its...
New Regional Leader for the Western Europe region has been elected!
We are absolutely thrilled to announce that Nigel Deekes, a dedicated member of the CML UK Facebook Group, has been elected as the temporary Regional Leader for the Western Europe region in our...
HARMONY Alliance | The three new patient-initiated CML research projects
The patient community contributing to the HARMONY Alliance has worked hard on some companies and institutions to get them also provide quality of life and toxicity data from the clinical trials. It...


